Catalog # |
Size |
Price |
|
|---|---|---|---|
| 058-02 | 500 µg | $97 |
 )
)
|
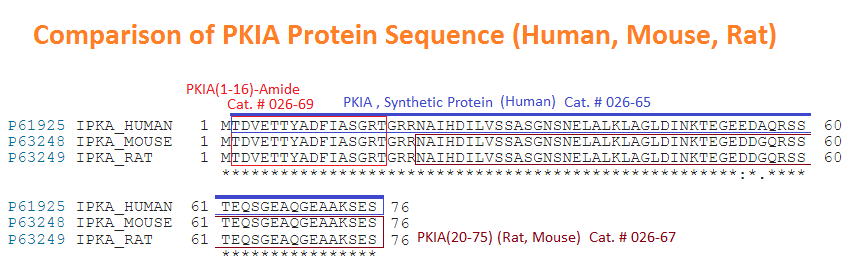
Thr-Thr-Tyr-Ala-Asp-Phe-Ile-Ala-Ser-Gly-Arg-Thr-Gly-Arg-Arg-Asn-Ala-Ile-His-Asp
|
| 2221.11 | |
|
| ≥ 95% |
|
| Exhibits correct molecular weight |
|
| Soluble in water |
|
|
Up to 6 months in lyophilized form at 0-5°C. For best results, rehydrate just before use. After rehydration, keep solution at +4°C for up to 5 days or freeze at -20°C for up to 3 months. Aliquot before freezing to avoid repeated freeze-thaw cycles. |
|
| White powder |
|
| Each vial contains 500 µg of NET peptide. |

G-protein-coupled-receptor (GPCR) signaling is exquisitely controlled to achieve spatial and temporal specificity. The endogenous protein kinase inhibitor peptide (PKI) confines the spatial and temporal spread of the activity of protein kinase A (PKA), which integrates inputs from three major types of GPCRs. Despite its wide usage as a pharmaceutical inhibitor of PKA, it was unclear whether PKI only inhibits PKA activity. Here, the effects of PKI on 55 mouse kinases were tested in in vitro assays. We found that in addition to inhibiting PKA activity, both PKI (6–22) amide and full-length PKIα facilitated the activation of multiple isoforms of protein kinase C (PKC), albeit at much higher concentrations than necessary to inhibit PKA. Thus, our results call for appropriate interpretation of experimental results using PKI as a pharmaceutical agent. Furthermore, our study lays the foundation to explore the potential functions of PKI in regulating PKC activity and in coordinating PKC and PKA activities.
Chen Y, Sabatini BL. The kinase specificity of protein kinase inhibitor peptide. Front Pharmacol. 2021;12:632815.
Abstract: The PKA-inhibitor (PKI) family members PKIα, PKIβ, and PKIγ bind with high affinity to PKA and block its kinase activity, modulating the extent, and duration of PKA-mediated signaling events. While PKA is a well-known regulator of physiological and oncogenic events, the role of PKI proteins in these pathways has remained elusive. Here, by measuring activation of the MAPK pathway downstream of GPCR-Gαs-cAMP signaling, we show that the expression levels of PKI proteins can alter the balance of activation of two major cAMP targets: PKA and EPAC. Our results indicate that PKA maintains repressive control over MAPK signaling as well as a negative feedback on cAMP concentration. Overexpression of PKI and its subsequent repression of PKA dysregulates these signaling pathways, resulting in increased intracellular cAMP, and enhanced activation of EPAC and MAPK. We also find that amplifications of PKIA are common in prostate cancer and are associated with reduced progression free survival. Depletion of PKIA in prostate cancer cells leads to reduced migration, increased sensitivity to anoikis and reduced tumor growth. By altering PKA activity PKI can act as a molecular switch, driving GPCR-Gαs-cAMP signaling toward activation of EPAC-RAP1 and MAPK, ultimately modulating tumor growth.
Hoy JJ, Salinas Parra N, Park J, Kuhn S, Iglesias?Bartolome R. Protein kinase A inhibitor proteins (Pkis) divert GPCR?Gαs?cAMP signaling toward EPAC and ERK activation and are involved in tumor growth. FASEB j. 2020;34(10):13900-13917.
The protein kinase enzyme family plays a pivotal role in almost every aspect of cellular function, including cellular metabolism, division, proliferation, transcription, movement, and survival. Protein kinase A (PKA), whose activation is triggered by cyclic adenosine monophosphate (cAMP), is widely distributed in various systems and tissues throughout the body and highly related to pathogenesis and progression of various kinds of diseases. The inhibition of PKA activation is essential for the study of PKA functions. Protein kinase inhibitor peptide (PKI) is a potent, heat-stable, and specific PKA inhibitor. It has been demonstrated that PKI can block PKA-mediated phosphorylase activation. Since then, researchers have a lot of knowledge about PKI. PKI is considered to be the most effective and specific method to inhibit PKA and is widely used in related research. In this review, we will first introduce the knowledge on the activation of PKA and mechanisms related on the inhibitory effects of PKI on PKA. Then, we will compare PKI-mediated PKA inhibition vs. several popular methods of PKA inhibition.
Liu C, Ke P, Zhang J, Zhang X, Chen X. Protein kinase inhibitor peptide as a tool to specifically inhibit protein kinase a. Front Physiol. 2020;11:574030
Abstract: Synthetic peptides corresponding to the active domain of the heat-stable inhibitor protein of cAMP-dependent protein kinase (Cheng, H.-C., Kemp, B. E., Pearson, R. B., Smith, A. J., Misconi, L., Van Patten, S. M., and Walsh, D. A. (1986) J. Biol. Chem. 261, 989-992) were tested as inhibitors of cGMP-dependent protein kinase. The peptides themselves were not substrates. cGMP-dependent protein kinase activity was assayed using histone H2B and two synthetic peptide substrates. Consistent with previous observations of other peptide inhibitors of this enzyme (Glass, D. B. (1983) Biochem. J. 213, 159-164), the inhibitory peptides had no effect on the phosphorylation of histone H2B, but they competitively inhibited cGMP-dependent phosphorylation of the two peptide substrates. The parent inhibitor peptide, PKI(5-24)amide, and a series of analogs had Ki (or IC50) values for cGMP-dependent protein kinase in the range of 15-190 microM. In contrast to their effects on the cAMP-dependent protein kinase, the inhibitory peptides were substantially less potent with cGMP-dependent protein kinase, and potency was reduced by the presence of the NH2-terminal residues (residues 5-13). We conclude that the two protein kinases share a recognition of the basic amino acid cluster within the pseudosubstrate region of the peptide, but that the cGMP-dependent protein kinase does not recognize additional NH2-terminal determinants that make the inhibitor protein extremely potent toward the cAMP-dependent enzyme. Even- when tested at high concentrations and with peptide substrates, the native inhibitor protein did not inhibit cGMP-dependent protein kinase under assay conditions in which the peptides derived from it were inhibitory. Thus, the native inhibitor protein appears to have structural features which block interaction with the cGMP-dependent enzyme and enhance its selectivity for cAMP-dependent protein kinase.
Glass DB, Cheng HC, Kemp BE, Walsh DA. Differential and common recognition of the catalytic sites of the cGMP-dependent and cAMP-dependent protein kinases by inhibitory peptides derived from the heat-stable inhibitor protein. Journal of Biological Chemistry. 1986;261(26):12166-12171.
No References
| Catalog# | Product | Size | Price | Buy Now |
|---|
Social Network Confirmation