

BACKGROUND: In vitro reactions are useful to identify putative enzyme substrates, but in vivo validation is required to identify actual enzyme substrates that have biological meaning. To investigate in vivo effects of prolyl endopeptidase (PREP), a serine protease, on α melanocyte stimulating hormone (α-MSH), we developed a new mass spectrometry based technique to quantitate, in multiplex, the various forms of α-MSH.METHODS: Using Multiple Reaction Monitoring (MRM), we analyzed peptide transitions to quantify three different forms of α-MSH. Transitions were first confirmed using standard peptides. Samples were then analyzed by mass spectrometry using a triple quadrupole mass spectrometer, after elution from a reverse phase C18 column by a gradient of acetonitrile.RESULTS: We first demonstrate in vitro that PREP digests biological active α melanocyte stimulating hormone (α-MSH1-13), by cleaving the terminal amidated valine and releasing a truncated α melanocyte stimulating hormone (α-MSH1-12) product - the 12 residues α-MSH form. We then use the technique in vivo to analyze the MRM transitions of the three different forms of α-MSH: the deacetylated α-MSH1-13, the acetylated α-MSH1-13 and the truncated form α-MSH1-12. For this experiment, we used a mouse model (PREP-GT) in which the serine protease, prolyl endopeptidase, is deficient due to a genetrap insertion. Here we report that the ratio between acetylated α-MSH1-13 and α-MSH1-12 is significantly increased (P-value = 0.015, N = 6) in the pituitaries of PREP-GT mice when compared to wild type littermates. In addition no significant changes were revealed in the relative level of α-MSH1-13 versus the deacetylated α-MSH1-13. These results combined with the demonstration that PREP digests α-MSH1-13 in vitro, strongly suggest that α-MSH1-13 is an in vivo substrate of PREP.CONCLUSION: The multiplex targeted quantitative peptidomics technique we present in this study will be decidedly useful to monitor several neuropeptide enzymatic reactions in vivo under varying conditions.
Perroud B, Alvarado RJ, Espinal GM, Morado AR, Phinney BS, Warden CH. Mol Brain. 2009;2:14.
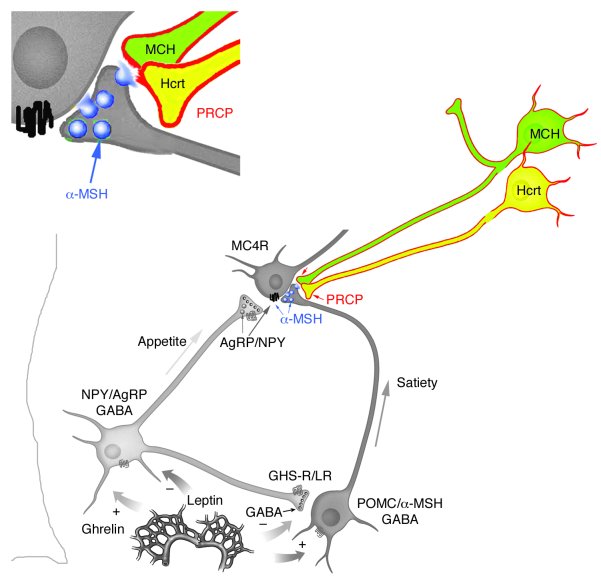
The anorexigenic neuromodulator α-melanocyte-stimulating hormone (α-MSH; referred to here as α-MSH1-13) undergoes extensive posttranslational processing, and its in vivo activity is short lived due to rapid inactivation. The enzymatic control of α-MSH1-13 maturation and inactivation is incompletely understood. Here we have provided insight into α-MSH1-13 inactivation through the generation and analysis of a subcongenic mouse strain with reduced body fat compared with controls. Using positional cloning, we identified a maximum of 6 coding genes, including that encoding prolylcarboxypeptidase (PRCP), in the donor region. Real-time PCR revealed a marked genotype effect on Prcp mRNA expression in brain tissue. Biochemical studies using recombinant PRCP demonstrated that PRCP removes the C-terminal amino acid of α-MSH1-13, producing α-MSH1-12, which is not neuroactive. We found that Prcp was expressed in the hypothalamus in neuronal populations that send efferents to areas where α-MSH1-13 is released from axon terminals. The inhibition of PRCP activity by small molecule protease inhibitors administered peripherally or centrally decreased food intake in both wild-type and obese mice. Furthermore, Prcp-null mice had elevated levels of α-MSH1-13 in the hypothalamus and were leaner and shorter than the wild-type controls on a regular chow diet; they were also resistant to high-fat diet-induced obesity. Our results suggest that PRCP is an important component of melanocortin signaling and weight maintenance via control of active α-MSH1-13 levels.
Wallingford N, Perroud B, Gao Q, et al. J Clin Invest. 2009;119(8):2291-303.
We report the isolation of a novel human circulating proopiomelanocortin-derived peptide named VA-ß-MSH from hemofiltrate and its pharmacological characterization. Screening for lipolytic activity in differentiated 3T3-L1 adipocytes led to the isolation from a hemofiltrate peptide library by alternating reversed-phase and cation-exchange chromatography. In the course of this isolation, we also identified human ß-MSH (1-22). We synthesized VA-ß-MSH by the Fmoc (N-(9-fluorenyl)-methoxycarbonyl) solid phase method and used synthetic ß-MSH (1-22) to confirm that both isolated peptides are lipolytically active in a dosedependent manner in differentiated 3T3-L1 adipocytes in the nanomolar range. Using cAMPELISA, we demonstrate that stimulation with both peptides caused a strong cAMP elevation in this cell system. Furthermore, we show that the selective inhibitors of cAMP-dependent protein kinase, Rp-8-CPT-cAMPS and H89, significantly reduce VA-ß-MSH- and ß-MSH (1-22)-mediated lipolysis. Although isolated here following its lipolytic activity on 3T3-L1 cells, this newly identified circulating human melanocortin may also serve other functions in human physiology. Moreover, the fact that these peptides have been identified following a functional assay but have been overseen in large proteomic approaches underscore the importance for such approaches in order to identify previously undescribed circulating bioactive molecules.
Fricke K., et al. Endocrinology. First published January 13, 2005 as doi:10.1210/en.2004-1097
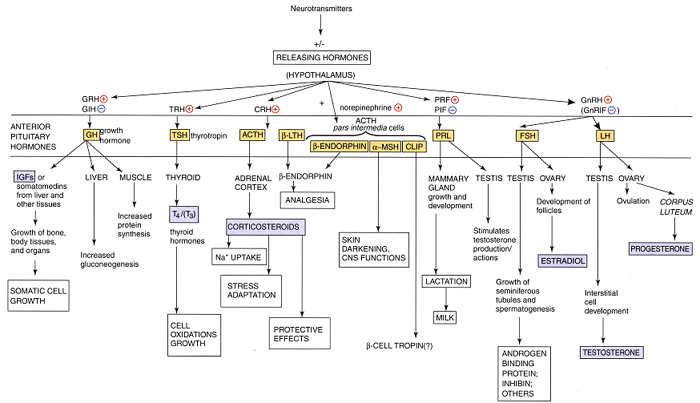
The roles of the melanocortin 3 receptor (MC3-R) and its agonist, γ(2)-melanocyte-stimulating hormone (γ(2)-MSH) in the regulation of the hypothalamo-pituitary-gonadal (HPG) axis are poorly understood. Here we show γ(2)-MSH stimulated intracellular cAMP accumulation and gonadotrophin-releasing hormone (GnRH) secretion in the immortalised GnRH cell line GT(1)-7. The MC3/4-R antagonist Agrp blocked these actions. Reverse transcriptase polymerase chain reaction demonstrated GT(1)-7 cells express MC3-R mRNA. γ(2)-MSH also stimulated GnRH release from hypothalamic explants. In vivo, γ(2)-MSH administration into the medial preoptic area significantly increased plasma luteinising hormone. MC3-R and γ(2)-MSH may modulate the HPG axis.
Stanley SA, et al. FEBS Lett. 2003 May 22;543(1-3):66-70
We report the isolation of a novel human circulating proopiomelanocortin-derived peptide named VA-ß-MSH from hemofiltrate and its pharmacological characterization. Screening for lipolytic activity in differentiated 3T3-L1 adipocytes led to the isolation from a hemofiltrate peptide library by alternating reversed-phase and cation-exchange chromatography. In the course of this isolation, we also identified human ß-MSH (1-22). We synthesized VA-ß-MSH by the Fmoc (N-(9-fluorenyl)-methoxycarbonyl) solid phase method and used synthetic ß-MSH (1-22) to confirm that both isolated peptides are lipolytically active in a dosedependent manner in differentiated 3T3-L1 adipocytes in the nanomolar range. Using cAMPELISA, we demonstrate that stimulation with both peptides caused a strong cAMP elevation in this cell system. Furthermore, we show that the selective inhibitors of cAMP-dependent protein kinase, Rp-8-CPT-cAMPS and H89, significantly reduce VA-ß-MSH- and ß-MSH (1-22)-mediated lipolysis. Although isolated here following its lipolytic activity on 3T3-L1 cells, this newly identified circulating human melanocortin may also serve other functions in human physiology. Moreover, the fact that these peptides have been identified following a functional assay but have been overseen in large proteomic approaches underscore the importance for such approaches in order to identify previously undescribed circulating bioactive molecules.
Fricke K., et al. Endocrinology. First published January 13, 2005 as doi:10.1210/en.2004-1097
α-Melanocyte stimulating hormone (MSH) has generally been assumed to be the endogenous ligand acting at the melanocortin-4 receptor (MC4-R), activation of which in the hypothalamus leads to reduced feeding. However, β-MSH is also capable of activating MC4-R and inhibiting feeding. Here, we investigated the possibility that β-MSH acts as an endogenous MC4-R agonist and that this melanocortin peptide plays a role in the regulation of feeding and energy balance. We found that β-MSH had significantly higher affinities than α-MSH at both human MC4-R transfected into CHO cells (K(i): β-MSH, 11.40.4 nmol/l versus α-MSH, 32416 nmol/l, P<0.001) and MC4-R in rat hypothalamic homogenates (K(i): β-MSH, 5.00.4 nmol/l versus α-MSH, 22.52.3 nmol/l, P<0.001). Incubation of brain slices with 5 microM β-MSH significantly increased [35S]GTPγS binding by 140-160% (P<0.001), indicating activation of G-protein-coupled receptors (GPCRs), in the hypothalamic ventromedial (VMH), dorsomedial (DMH), arcuate (ARC) and paraventricular (PVN) nuclei. These sites match the distribution of β-MSH immunoreactive fibres and also the distribution of MC4-R binding sites which we and others previously reported. Food-restriction significantly increased β-MSH levels in the VMH, DMH and ARC (all P<0.05) above freely-fed controls, whilst α-MSH concentrations were unchanged. We propose that increased β-MSH concentrations reflect blockade of the peptide's release in these sites, consistent with the increased hunger and the known up-regulation of MC4-R in the same nuclei. Thus, we conclude that (1). β-MSH has higher affinity at MC4-R than α-MSH; (2). β-MSH activates GPCR in these sites, which are rich in MC4-R; and (3). β-MSH is present in hypothalamic nuclei that regulate feeding and its concentrations alter with nutritional state. We suggest that β-MSH rather than α-MSH is the key ligand at the MC4-R populations that regulate feeding, and that inhibition of tonic release of β-MSH is one mechanism contributing to hunger in under-feeding.
Harrold JA, et al. Peptides. 2003 Mar;24(3):397-405
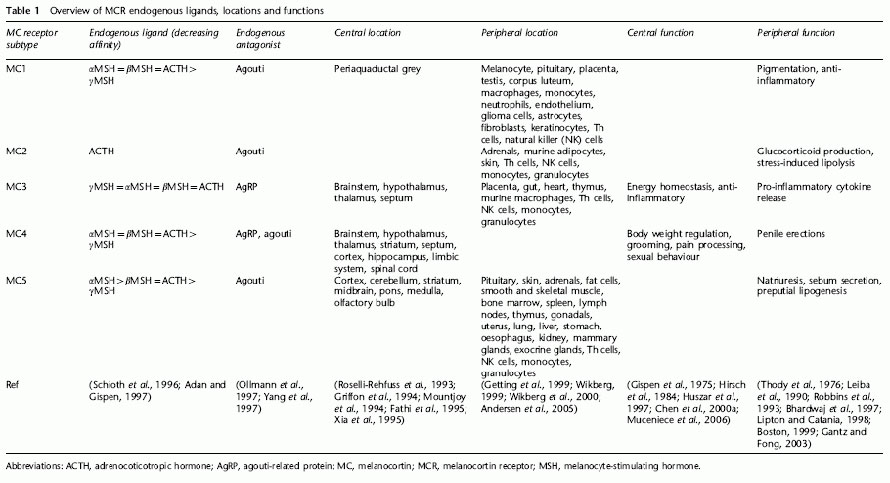
Overview of MCR endogenous ligands, locations and functions
| Catalog# | Product | Standard Size | Price |
|---|---|---|---|
| 043-01 | MSH, alpha (Human, Rat, Mouse) | 500 µg | $48 |
| EK-043-01 | MSH, alpha (Human, Rat, Mouse) - EIA Kit | 96 wells | $570 |
| FEK-043-01 | MSH, alpha (Human, Rat, Mouse) - Fluorescent EIA Kit | 96 wells | $624 |
| 043-06 | [Nle4, D-Phe7]-MSH, alpha (NDP-alpha-MSH) | 500 µg | $55 |
| H-043-01 | MSH, alpha (Human, Rat, Mouse) - Antibody | 50 µl | $225 |
| RK-043-01 | MSH, alpha (Human, Rat, Mouse) - RIA Kit | 125 tubes | $880 |
| EK-043-01CE | MSH, alpha (Human, Rat, Mouse) - EIA Kit, CE Mark Certified | 96 wells | $595 |
| FEK-043-01CE | MSH, alpha (Human, Rat, Mouse) - Fluorescent EIA Kit, CE Mark Certified | 96 wells | $649 |
| 043-20 | MSH Release Inhibiting Factor (MIF) | 5 mg | $35 |
| 043-42 | MSH, VA-beta (Human) | 100 µg | $167 |
Social Network Confirmation