
Ostn blocks the clearance action of NPR-C thereby increasing activity of the NPs in the bone compartment (A) Both the GC-B and NPR-C receptors are expressed in osteoblasts and chondrocytes, thus the activity of the NPs (normally CNP in the skeleton) is governed by the distribution of CNP between the signal mediating GC-B receptor and the NPR-C clearance receptor. (B) In the presence of Ostn, CNP access to NPR-C is blocked leading to increased binding of CNP to the GC-B receptor. This results in an increase in GC-B-mediated cGMP production, magnifying the downstream biological effects of the natriuretic system, which in the skeleton leads to elongated bones.
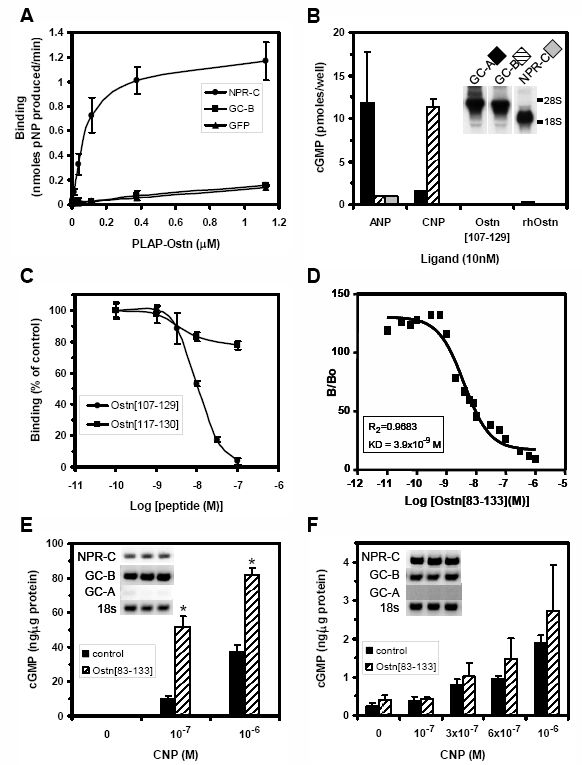

Ostn-TG mice with over-expression of Ostn in osteoblast lineage-specific cells were generated using the 3.6kb collagen type I promoter. Immunohistochemistry using an Ostn-specific antibody on 4-day-old tibia of wild type (WT) (A) and Ostn-TG (B) mice. Ostn expression in WT is non detectable (A) whereas overexpression in Ostn-TG is evident (red staining) on cuboidal osteoblasts adjacent to trabecular bone of the primary spongiosa (arrows), and within the perichondrium (arrowheads) (B). Counter stain is methylgreen. Scale bar is 200μm. (C, D) Gross appearance of WT (C) and Ostn-TG (D) which have a marked kyphosis presumably due to vertebral overgrowth. (E-F) Proliferation in growth plate chondrocytes was measured in tibiae of 7- week old male mice using PCNA immunohistochemistry. Proliferating chondrocytes are clearly more abundant in Ostn-TG (F) mice than WT littermates (E). Tail (G) and femur (H) lengths of 8-week old Ostn-TG line 650 males are significantly longer than wildtype littermates (n=7-12). (I) Quantification of PCNA immunohistochemistry showed an 80% increase in staining of growth plate chondrocytes (n=4) (J). cGMP levels were significantly higher in 10-14 day-old Ostn-TG mice femurs and tibiae than their WT littermates (n=11-28). *p<0.05; **p<0.01, Ostn-TG vs. WT. Data are expressed as mean ± SE. Comparison were made by ANOVA using Statview.

A, in situ localization of osteocrin transcripts in e16.5 mouse ribs. Osteocrin is localized within a subset of Cbfa-1-positive cells in a subperiosteal layer (per). Active mature osteoblasts are signified by an arrowhead (x200). B, expression of osteocrin in whole e17.5 fore limb. Osteocrin is clearly localized in the osteoblasts on the periphery of the bone (x100). C, in e16.5 mouse tibiae, osteocrin co-localizes with a subset of Cbfa-1-positive cells, exclusive from the more mature osteocalcin-positive cells found on the bone surface and in the trabecular bone (arrowheads) (x400).
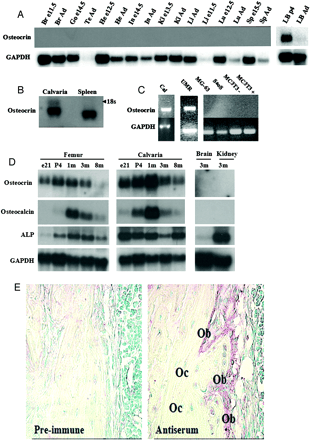
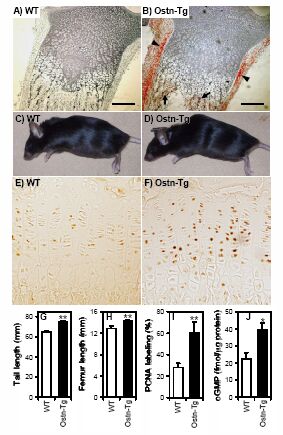
A, Northern blot showing bone specificity of mouse osteocrin expression. Osteocrin is only detected in neonate and adult long bones and calvaria. No message was detected in embryonic (e) or adult (ad) non-bone tissues. Five µg total RNA was loaded. Br, brain; Go, gonads; Te, testes; He, heart; In, intestine; Ki, kidney; Li, liver; Lu, lung; Sp, spleen. B, osteocrin in detected by Northern blot in rat spleen. The transcript size of adult rat spleen osteocrin is smaller than that for adult rat calvaria. Fifteen µg of total RNA was loaded. The position of the 18 S ribosomal RNA band is marked. C, osteocrin expression was detected in embryonic calvariae and UMR106 cells by RT-PCR. No expression was detected in MG-63, SaoS-2, undifferentiated (-), or differentiated (+) MC3T3.E1 cells. D, Northern blot showing osteocrin expression in femora and calvariae in embryonic (e21), newborn (p4), growing (1 month, 1m), adult (3 month, 3m), and aged (8-month, 8m) rats. In femora, osteocrin expression was highest in newborn rats decreasing significantly in aged rats. Osteocrin expression peaks at 1-month in calvaria with a less marked decrease in older rats. Twenty-five µg of total RNA was loaded. ALP, alkaline phosphatase. For Figs. 4, A-D, GAPDH represents a loading control. E, immunohistochemistry with an osteocrin-specific antibody showing localization of osteocrin protein to active osteoblasts (Ob) in adult mouse tibia. The protein is absent from the mature osteocytes (Oc). No staining is visible in the sections stained with pre-immune serum (x400).
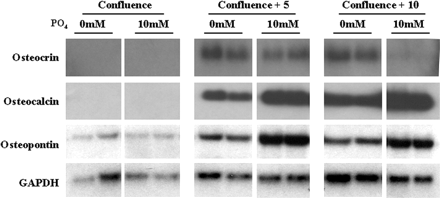
Northern blot of a time course of calvarial primary osteoblast differentiation at confluence and 5 and 10 days post-confluence in the presence and absence of 10 mM GP (PO4). Osteocrin is expressed at 5 and 10 days post-confluence but not at confluence. Cultures maintained in 10 mM GP until 10 days post-confluence exhibit a marked down-regulation in osteocrin expression. Osteocalcin and osteopontin expression are highest at 10 days post-confluence and are increased by GP treatment. Twenty µg of total RNA was loaded. GAPDH represents a loading control. Duplicate samples are shown.GP (PO4). Osteocrin is expressed at 5 and 10 days post-confluence but not at confluence. Cultures maintained in 10 mM GP until 10 days post-confluence exhibit a marked down-regulation in osteocrin expression. Osteocalcin and osteopontin expression are highest at 10 days post-confluence and are increased by GP treatment. Twenty µg of total RNA was loaded. GAPDH represents a loading control. Duplicate samples are shown.
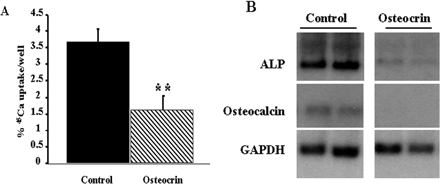
Primary calvarial osteoblastic cultures were treated with conditioned media from osteocrin or empty vector (control) transfected HEK293A cells from day 2 to 10 days post-confluence. A, 45Ca uptake was reduced 60% by osteocrin conditioned-media. Data is expressed as the mean ± S.E. of the % 45Ca incorporated into the cultures. *, p < 0.01. B, Northern blot demonstrating total repression of osteocalcin and a marked reduction in alkaline phosphatase expression in osteocrin treated cultures. Fifteen µg of total RNA was loaded. GAPDH represents a loading control. Duplicate samples are shown.
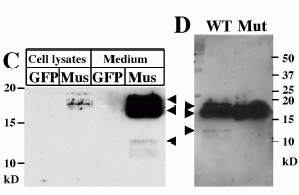
C, Secretion of musclin protein. The cell lysates and culture medium from C2C12 myocytes infected by retrovirus carrying Flag-tagged mouse musclin or GFP were immunoprecipitated, and subjected to western blot analysis, using anti-Flag antibody. Mus; musclin. D, Secretion of wild type and mutant musclin from HEK 293 cells. The culture medium from HEK 293 cells transfected with pEF-BOS-WT musclin Flag (left lane) or pEFBOS-Mut musclin Flag (76KKKR79-76A) (right lane) was subjected to immunoprecipitation followed by western blotting using anti-Flag antibody.
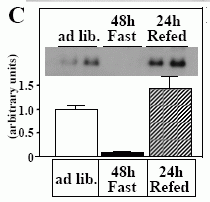
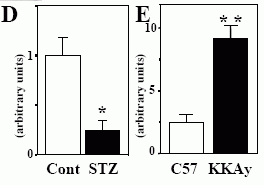
In vivo expression of musclin mRNA. A, Northern blot analysis of musclin expression in mice. B, Quantitative RT-PCR analysis of musclin mRNA expression in mice. C, Regulation of musclin mRNA expression by nutritional status. Gastrocnemius muscles of mice fed ad libitum, fasted for 48 h, or fasted for 48 h and refed for 24 h (n=5, each) were used for real-time RT-PCR. Inset: representative Northern blotting (n=2, each). D, Musclin mRNA expression in insulin-deficient STZ mice. Gastrocnemius muscles of STZ and control mice (n=5, each) were subjected to real-time RT-PCR. The values were expressed relative to the level of 36B4 mRNA. E, Musclin mRNA expression in KKAy mice. Gastrocnemius muscles of 13-wk-old KKAy mice and age-matched C57BL/6J mice (n=8, each) were used for real-time RT-PCR. The values were normalized to the level of cyclophilin mRNA. Data are mean +- SEM values. *p< 0.05, **p< 0.01.

Regulation of musclin mRNA expression by various hormones, and effects of musclin on insulin-stimulated glucose uptake and glycogen synthesis in C2C12 myocytes. A, Musclin mRNA expression during myocyte differentiation. C2C12 cells were harvested at indicated days after differentiation-induction for real-time PCR. B-D, Regulation of musclin mRNA expression by insulin (INS), IGF-1 (IGF), epinephrine (Epi), isoproterenol (Iso), and forskolin (FSK) (real-time PCR). The expression level was expressed relative to that of cyclophlin mRNA (n=3, each). E-F, Effect of recombinant musclin on 2-DG uptake and glycogen synthesis. After 5-h pre-treatment with Flag-peptide (0.5 ug/ml) as control or Flagtagged musclin (0.5 ug/ml), 2-DG uptake (E) and glycogen synthesis (F) were determined as described in Experimental Procedures. Data are mean+- SEM values (n=6). **p<0.01. Similar results were obtained in two other independent experiments.



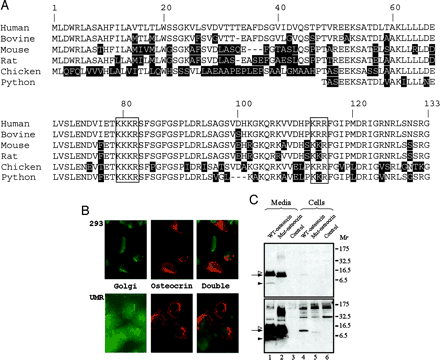











Social Network Confirmation