
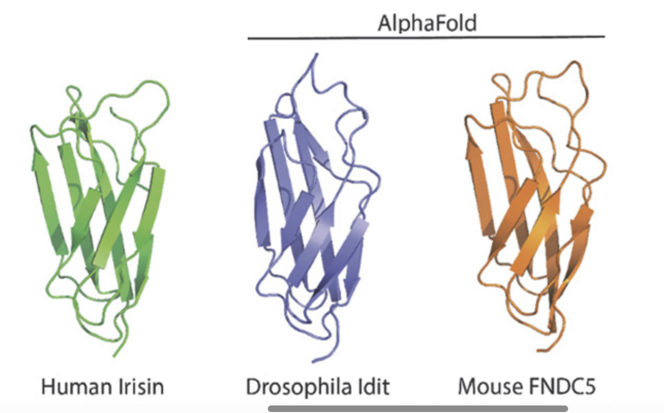
Image is from Proc Natl Acad Sci U S A. 2023 Sep 26;120(39):e2220556120.
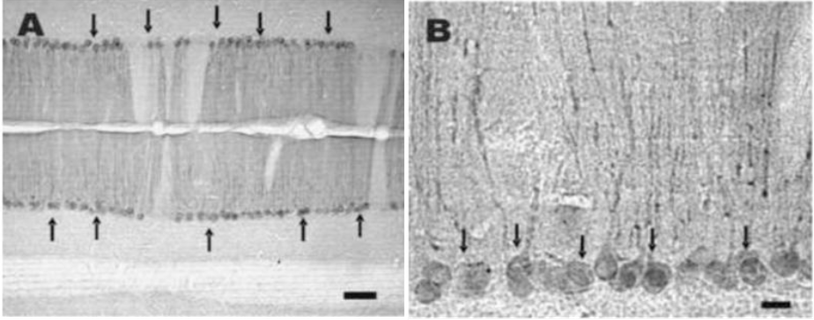
Cross sections of mouse cardiac and skeletal muscle fibers labeled with irisin antiserum. A and B, cardiac and skeletal muscle cells express varying intensities of irIRN. In skeletal muscle fibers, irIRN appears to be more densely localized to the cell membrane, with lower immunoreactivity close to the middle of muscle cells. Scale bar: 50 μm. Picture from Https://pubmed.ncbi.nlm.nih.gov/23470775/
Abstract: Mammalian FNDC5 encodes a protein precursor of Irisin, which is important for exercise-dependent regulation of whole-body metabolism. In a genetic screen in Drosophila, we identified Iditarod (Idit), which shows substantial protein homology to mouse and human FNDC5, as a regulator of autophagy acting downstream of Atg1/Atg13. Physiologically, Idit-deficient flies showed reduced exercise performance and defective cold resistance, which were rescued by exogenous expression of Idit. Exercise training increased endurance in wild-type flies, but not in Idit-deficient flies. Conversely, Idit is induced upon exercise training, and transgenic expression of Idit in wild-type flies increased endurance to the level of exercise trained flies. Finally, Idit deficiency prevented both exercise-induced increase in cardiac Atg8 and exercise-induced cardiac stress resistance, suggesting that cardiac autophagy may be an additional mechanism by which Idit is involved in the adaptive response to exercise. Our work suggests an ancient role of an Iditarod/Irisin/FNDC5 family of proteins in autophagy, exercise physiology, and cold adaptation, conserved throughout metazoan species.
Cobb T, Hwang I, Soukar M, et al. iditarod , a drosophila homolog of the irisin precursor fndc5 , is critical for exercise performance and cardiac autophagy. Proc Natl Acad Sci USA. 2023;120(39):e2220556120.
Abstract: Irisin is a recently identified myokine secreted from the muscle in response to exercise. In the rats and mice, immunohistochemical studies with an antiserum against irisin peptide fragment (42-112), revealed that irisin-immunoreactivity (irIRN) was detected in three types of cells; namely, skeletal muscle cells, cardiomyocytes, and Purkinje cells of the cerebellum. Tissue sections processed with irisin antiserum pre-absorbed with the irisin peptide (42-112) (1 μg/ml) showed no immunoreactivity. Cerebellar Purkinje cells were also immunolabeled with an antiserum against fibronectin type II domain containing 5 (FNDC5), the precursor protein of irisin. Double-labeling of cerebellar sections with irisin antiserum and glutamate decarboxylase (GAD) antibody showed that nearly all irIRN Purkinje cells were GAD-positive. Injection of the fluorescence tracer Fluorogold into the vestibular nucleus of the rat medulla retrogradely labeled a population of Purkinje cells, some of which were also irIRN. Our results provide the first evidence of expression of irIRN in the rodent skeletal and cardiac muscle, and in the brain where it is present in GAD-positive Purkinje cells of the cerebellum. Our findings together with reports by others led us to hypothesize a novel neural pathway, which originates from cerebellum Purkinje cells, via several intermediary synapses in the medulla and spinal cord, and regulates adipocyte metabolism.
The authors used Phoenix's Irisin antibody (Cat. #H-067-17) for the studies in this article.
Dun SL, Lyu RM, Chen YH, Chang JK, Luo JJ, Dun NJ. Irisin-immunoreactivity in neural and non-neural cells of the rodent. Neuroscience. 2013;240:155-162.
| Catalog# | Product | Standard Size | Price |
|---|---|---|---|
| 067-40 | Idit (19-97)-amide | 100ug | $395 |
Social Network Confirmation